Improved Processing of Buccal Swabs and Punches for Direct Amplification
Promega Corporation
Publication Date: 2012
Introduction
FTA® cards are used by many database laboratories as a way of providing long-term storage of blood or buccal samples at room temperature. Chemicals within these cards lyse cells, inactivate bacteria and make DNA available for subsequent amplification. However, these same chemicals can inhibit amplification unless they are first removed by extensive washing procedures. Recently, Promega launched several direct-amplification-compatible STR multiplexes that are tolerant of these chemicals (i.e., the PowerPlex® 18D, 21 and Y23 Systems) and allow direct amplification without prior washing (1) (2) (3) . While FTA® cards are widely used, they are not the sole solid support available to labs for storing blood or buccal samples. Buccal swabs may be obtained with simple cotton swabs or paper swabs like OmniSwab™ swabs. In addition, various nonFTA options exist for storing buccal cells such as S&S 903 paper or the Bode Buccal DNA Collector™ device. Blood also can be spotted directly onto S&S 903 paper for long-term storage. Unlike FTA® cards, these supports are not impregnated with chemicals that lyse cells, denature proteins and make DNA available for amplification. For these solid supports, a pretreatment step is required to ensure lysis and release of amplifiable DNA. Buccal swabs pose an additional obstacle in that they are not amenable to punching.
To help address these issues and allow direct amplification from buccal swabs and nonFTA and FTA® card punches using most PowerPlex® Systems, Promega offers the SwabSolution™, PunchSolution™ and AmpSolution™ suite of products.
The SwabSolution™ Kit (Cat.# DC8271) provides a simple and rapid method to generate an extract from buccal swabs that can be added directly to PowerPlex® reactions. The PunchSolution™ Kit (Cat.# DC9271) is used to pretreat nonFTA card punches prior to direct amplification using PowerPlex® Systems. The SwabSolution™ Kit and PunchSolution™ Kit both contain the 5X AmpSolution™ Reagent, which allows direct amplification of DNA from swab extracts and blood and buccal samples on FTA® and nonFTA storage card punches using the “AmpSolution™-dependent” PowerPlex® Systems listed in Table 1. A compatibility matrix of various PowerPlex® Systems for direct amplification from buccal swab extracts and nonFTA and FTA® card punches is shown in Table 1.
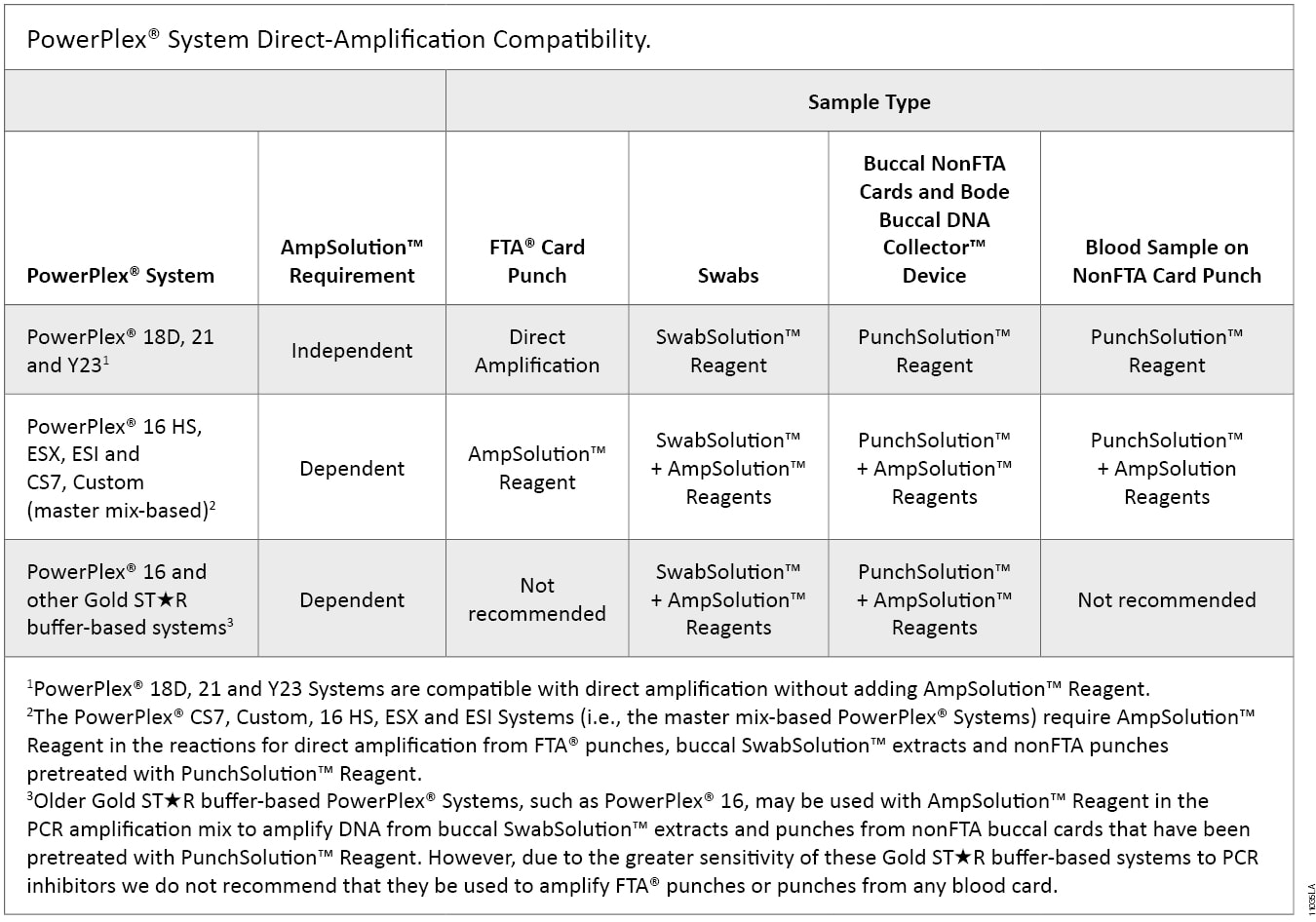 Table 1. PowerPlex® System Direct-Amplification Compatibility.
Table 1. PowerPlex® System Direct-Amplification Compatibility. SwabSolution™ Kit
The SwabSolution™ Kit contains both SwabSolution™ Reagent and 5X AmpSolution™ Reagent. The SwabSolution™ Reagent is added directly to a tube or well of a 2.2ml Square-Well Deep Well Plate (Cat.# V6781) containing a buccal swab (4) . Swabs are incubated for 30 minutes in a dry block set at 70°C (tube format) or on a dry block set at 90°C (plate format). See Figure 1. Following incubation, 2µl of this extract is added to 23µl of PCR amplification mix, and thermal cycling is performed using the recommended program and cycle number for the PowerPlex® System being used (4) . If an AmpSolution™-dependent PowerPlex® System is used, the 5X AmpSolution™ Reagent is included in the PCR amplification mix at a final concentration of 1X. Results obtained with SwabSolution™ extracts are comparable to those obtained with DNA purified using the DNA IQ™ System (Figure 2).
 Figure 1. SwabSolution™ workflow. The buccal swab is added to a tube or well of a 2.2ml Square-Well Deep Well Plate, and 1ml of SwabSolution™ Reagent is added. Samples are incubated for 30 minutes at 70°C (tube format) or 90°C (plate format). For the plate format the 2.2ml Square-Well Deep Well Plate is placed on a Heat Transfer Plate (Cat.# A2661) sitting on a dry block set to 90°C. This ensures contents of wells reach 70°C. Following incubation, 2µl is used per 25µl amplification reaction.
Figure 1. SwabSolution™ workflow. The buccal swab is added to a tube or well of a 2.2ml Square-Well Deep Well Plate, and 1ml of SwabSolution™ Reagent is added. Samples are incubated for 30 minutes at 70°C (tube format) or 90°C (plate format). For the plate format the 2.2ml Square-Well Deep Well Plate is placed on a Heat Transfer Plate (Cat.# A2661) sitting on a dry block set to 90°C. This ensures contents of wells reach 70°C. Following incubation, 2µl is used per 25µl amplification reaction. Figure 2. PowerPlex® 18D profile of SwabSolution™ extract and DNA IQ™-purified DNA. Duplicate OmniSwab™ samples from the same individual were extracted with 1ml of SwabSolution™ Reagent or purified using the DNA IQ™ System and eluted in 100µl of DNA IQ™ Elution Buffer, as described in the DNA IQ™ System—Database Protocol Technical Bulletin TB297. PowerPlex® 18D amplification reactions (25µl) were assembled using 2µl of SwabSolution™ extract or 1µl of DNA IQ™-purified DNA. Amplification reactions were performed using the recommended PowerPlex® 18D cycling conditions for 27 cycles on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection).
Figure 2. PowerPlex® 18D profile of SwabSolution™ extract and DNA IQ™-purified DNA. Duplicate OmniSwab™ samples from the same individual were extracted with 1ml of SwabSolution™ Reagent or purified using the DNA IQ™ System and eluted in 100µl of DNA IQ™ Elution Buffer, as described in the DNA IQ™ System—Database Protocol Technical Bulletin TB297. PowerPlex® 18D amplification reactions (25µl) were assembled using 2µl of SwabSolution™ extract or 1µl of DNA IQ™-purified DNA. Amplification reactions were performed using the recommended PowerPlex® 18D cycling conditions for 27 cycles on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection).DNA extracted from buccal swabs using the SwabSolution™ Reagent is stable in the extracts for at least 12 months at 4°C, providing the ability to re-amplify the same extracts in the future. The SwabSolution™ Reagent maximizes DNA recovery from a buccal swab but does not normalize DNA yield. If desired, you can quantitate DNA in the extracts by fluorescent double-stranded-DNA-binding-dye-based quantitation assays such as the QuantiFluor™ dsDNA System (Cat.# E2670) or Quant-iT™ PicoGreen® dsDNA Reagent (Life Technologies). Both of these fluorescent quantitation assays tolerate SwabSolution™ extract at up to 5% of the total reaction volume. Alternatively, DNA in these extracts may be quantitated using the Plexor® HY System (Cat.# DC1000 and DC1001) if 5X AmpSolution™ Reagent is included in the real-time amplification reaction at a final concentration of 1X (4) (5) . DNA concentrations obtained by these various methods are comparable (Table 2) and may be used for normalizing input template amounts.
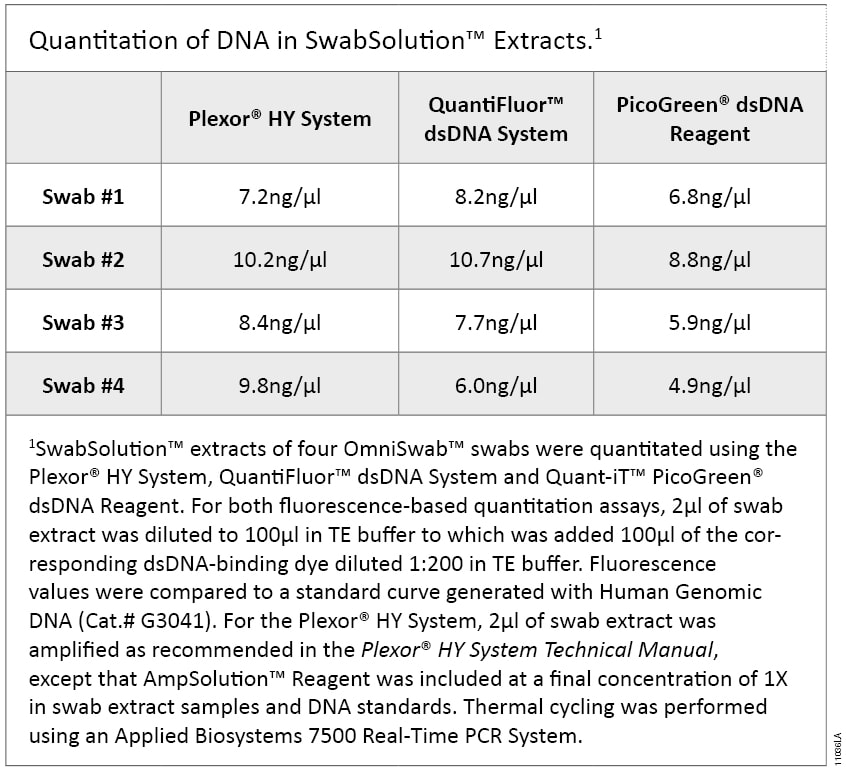 Table 2. Quantitation of DNA in SwabSolution™ Extracts.
Table 2. Quantitation of DNA in SwabSolution™ Extracts. PunchSolution™ Kit
The PunchSolution™ Kit contains PunchSolution™ Reagent and 5X AmpSolution™ Reagent. PunchSolution™ Reagent is used to pretreat 1.2mm punches from blood or buccal samples on nonFTA cards (4) (Figure 3). Pretreatment occurs in the same PCR plate used for amplification. The volume of PunchSolution™ Reagent added (10µl) is sufficient to cover the bottom of the well, making it possible to add punches before or after reagent addition. The punches then are incubated at 70°C for 30 minutes in a thermal cycler or dry block capable of accepting a PCR plate. The plate is not sealed during this incubation so that the PunchSolution™ Reagent evaporates completely. After this incubation, 25µl of PowerPlex® PCR amplification mix is added to the well containing the dried punch. Thermal cycling is performed using the recommended program and cycle number for the PowerPlex® System being used (4) . If an AmpSolution™-dependent PowerPlex® System is being used, AmpSolution™ Reagent is included in the PCR amplification mix at a final concentration of 1X. Figure 4 illustrates the effectiveness of this pretreatment to generate a profile with the PowerPlex® 18D System from a 1.2mm punch of a Bode Buccal DNA Collector™ device. Following processing with PunchSolution™ Reagent and amplification, a robust profile was generated, whereas amplification from an untreated punch taken adjacent to the treated punch on the same collector resulted in very poor signal (Figure 4). A 100% first-pass success rate was observed when using the PowerPlex® 18D System to amplify 80 separate pretreated punches from Bode Buccal DNA Collector™ devices and 80 separate pretreated punches from blood on S&S 903 paper (data not shown).
 Figure 3. PunchSolution™ workflow. A 1.2mm punch from a nonFTA card is added to the well of a PCR plate, and 10µl of PunchSolution™ Reagent is added. Samples are incubated at 70°C for 30 minutes with an open lid to allow evaporation. Following incubation, 25µl of PCR amplification mix is added, the plate is sealed and thermal cycling is performed as recommended for the PowerPlex® System being used.
Figure 3. PunchSolution™ workflow. A 1.2mm punch from a nonFTA card is added to the well of a PCR plate, and 10µl of PunchSolution™ Reagent is added. Samples are incubated at 70°C for 30 minutes with an open lid to allow evaporation. Following incubation, 25µl of PCR amplification mix is added, the plate is sealed and thermal cycling is performed as recommended for the PowerPlex® System being used. Figure 4. Effect of PunchSolution™ treatment on generation of a PowerPlex® 18D profile. A 1.2mm punch from a Bode Buccal DNA Collector™ device (Bode Technology Group, Inc.) was incubated with 10µl of PunchSolution™ Reagent at 70°C for 30 minutes prior to addition of 25µl of PowerPlex® 18D PCR amplification mix (Panel A) or left untreated (Panel B). Each experimental condition was performed in quadruplicate with punches taken from adjacent regions of the same Bode Buccal DNA Collector™ device. Amplification reactions were performed using the recommended PowerPlex® 18D cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). A representative electropherogram is shown for PunchSolution™-treated and untreated punches.
Figure 4. Effect of PunchSolution™ treatment on generation of a PowerPlex® 18D profile. A 1.2mm punch from a Bode Buccal DNA Collector™ device (Bode Technology Group, Inc.) was incubated with 10µl of PunchSolution™ Reagent at 70°C for 30 minutes prior to addition of 25µl of PowerPlex® 18D PCR amplification mix (Panel A) or left untreated (Panel B). Each experimental condition was performed in quadruplicate with punches taken from adjacent regions of the same Bode Buccal DNA Collector™ device. Amplification reactions were performed using the recommended PowerPlex® 18D cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). A representative electropherogram is shown for PunchSolution™-treated and untreated punches.5X AmpSolution™ Reagent
The 5X AmpSolution™ Reagent is available as part of the SwabSolution™ and PunchSolution™ Kits or as a separate product (Cat.# DM1231). The reagent is added to AmpSolution™-dependent PowerPlex® Systems (Table 1) to enable direct amplification from SwabSolution™ extracts and nonFTA punches pretreated with PunchSolution™ Reagent. Without addition of 5X AmpSolution™ Reagent, these PowerPlex® Systems will fail to generate a profile (Figure 5). In addition, adding 5X AmpSolution™ Reagent to the PCR amplification mix of “master mix”-based, AmpSolution™-dependent PowerPlex® Systems (see Table 1) enables direct amplification from one blood punch or two buccal FTA® punches (Figure 6). Thus, addition of the 5X AmpSolution™ Reagent makes these PowerPlex® Systems capable of direct amplification from unwashed FTA® punches, similar to the PowerPlex® 18D, 21 and Y23 Systems.
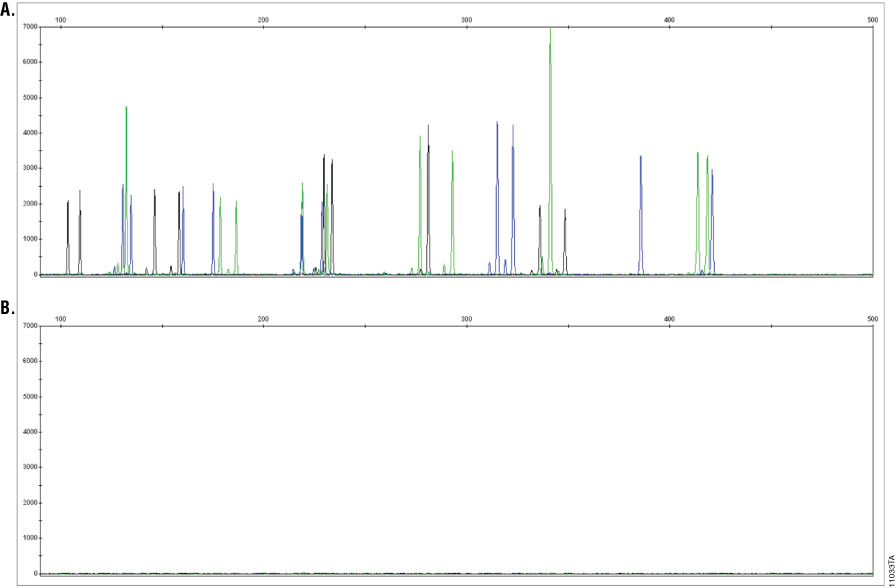 Figure 5. AmpSolution™ dependency of PowerPlex® 16 HS System with PunchSolution™-treated 1.2mm punches. Each punch from a Bode Buccal DNA Collector™ device was incubated with 10µl of PunchSolution™ Reagent at 70°C for 30 minutes prior to addition of 25µl of PowerPlex® 16 HS PCR amplification mix in the presence (Panel A) or absence (Panel B) of AmpSolution™ Reagent at a final concentration of 1X. Each experimental condition was performed in quadruplicate with punches taken from adjacent regions of the same Bode Buccal DNA Collector™ device. Amplification reactions were performed using the recommended PowerPlex® 16 HS cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). A representative electropherogram is shown for each.
Figure 5. AmpSolution™ dependency of PowerPlex® 16 HS System with PunchSolution™-treated 1.2mm punches. Each punch from a Bode Buccal DNA Collector™ device was incubated with 10µl of PunchSolution™ Reagent at 70°C for 30 minutes prior to addition of 25µl of PowerPlex® 16 HS PCR amplification mix in the presence (Panel A) or absence (Panel B) of AmpSolution™ Reagent at a final concentration of 1X. Each experimental condition was performed in quadruplicate with punches taken from adjacent regions of the same Bode Buccal DNA Collector™ device. Amplification reactions were performed using the recommended PowerPlex® 16 HS cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). A representative electropherogram is shown for each. Figure 6. AmpSolution™ Reagent enables direct amplification from buccal samples on 1.2mm FTA® punches using the PowerPlex® 16 HS System. Two 1.2mm punches from a buccal FTA® card were added to 25µl of PowerPlex® 16 HS PCR amplification mix in the presence (Panel A) or absence (Panel B) of AmpSolution™ Reagent at a final concentration of 1X. Amplification reactions were performed using the recommended PowerPlex® 16 HS cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). Amplifications were performed in quadruplicate for both experimental conditions using punches taken from adjacent regions of the FTA® card. A representative electropherogram is shown for each.
Figure 6. AmpSolution™ Reagent enables direct amplification from buccal samples on 1.2mm FTA® punches using the PowerPlex® 16 HS System. Two 1.2mm punches from a buccal FTA® card were added to 25µl of PowerPlex® 16 HS PCR amplification mix in the presence (Panel A) or absence (Panel B) of AmpSolution™ Reagent at a final concentration of 1X. Amplification reactions were performed using the recommended PowerPlex® 16 HS cycling conditions for 27 cycles (10/17) on a GeneAmp® PCR System 9700 thermal cycler. Amplified products were separated on an Applied Biosystems® 3130 Genetic Analyzer (3kV, 5-second injection). Amplifications were performed in quadruplicate for both experimental conditions using punches taken from adjacent regions of the FTA® card. A representative electropherogram is shown for each.Conclusion
The SwabSolution™ Kit provided a quick and simple method for extracting amplifiable DNA that can be added directly to PowerPlex® Systems or stored for future amplification. If desired, the amount of DNA in the extracts can be quantitated using fluorescent methods or real-time qPCR methods, and these quantitation values can be used for normalizing input template amounts. The PunchSolution™ Kit allowed amplification of DNA from nonFTA punches and resulted in a significant increase in signal over untreated punches and a high first-pass success rate in generating single-source DNA profiles.
The 5X AmpSolution ™ Reagent enabled direct amplification of DNA from SwabSolution™ extracts, PunchSolution™-treated nonFTA punches and untreated FTA® punches using many of the PowerPlex® Systems that were not originally designed to be compatible with direct amplification.
In summary, this suite of products increases the number of sample types that can be used with PowerPlex® Systems.
Related Protocols
- SwabSolution™ Kit Technical Manual
- PunchSolution™ Kit Technical Manual
- PowerPlex® Y23 System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- PowerPlex® 21 System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- PowerPlex® 18D System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
Article References
- Oostdik, K. et al. (2011) The PowerPlex® 18D System: A direct-amplification STR system with reduced thermal cycling time. Profiles in DNA.
- Ensenberger, M.G. et al. (2012) Development of the PowerPlex® 21 System. Profiles in DNA.
- Thompson, J. et al. (2012) The PowerPlex® Y23 System: A new Y-STR multiplex for casework and database applications Profiles in DNA.
- Plexor® HY System Technical Manual for the Applied Biosystems 7500 and 7500 FAST Real-Time PCR Systems TM293, Promega Corporation.
How to Cite This Article
Scientific Style and Format, 7th edition, 2006
McLaren, R.S. et al. Improved Processing of Buccal Swabs and Punches for Direct Amplification. [Internet] 2012. [cited: year, month, date]. Available from: https://www.promega.com/resources/profiles-in-dna/2012/improved-processing-of-buccal-swabs-and-punches-for-direct-amplification/
American Medical Association, Manual of Style, 10th edition, 2007
McLaren, R.S. et al. Improved Processing of Buccal Swabs and Punches for Direct Amplification. Promega Corporation Web site. https://www.promega.com/resources/profiles-in-dna/2012/improved-processing-of-buccal-swabs-and-punches-for-direct-amplification/ Updated 2012. Accessed Month Day, Year.
Contribution of an article to Profiles in DNA does not constitute an endorsement of Promega products.
Plexor and PowerPlex are registered trademarks of Promega Corporation. AmpSolution, DNA IQ, PunchSolution, QuantiFluor and SwabSolution are trademarks of Promega Corporation.
Applied Biosystems is a registered trademark of Applied Biosystems. Bode Buccal DNA Collector is a trademark of the Bode Technology Group, Inc. FTA is a registered trademark of Flinders Technologies, Pty, Ltd., and is licensed to Whatman. GeneAmp is a registered trademark of Roche Molecular Systems, Inc. OmniSwab is a trademark of Whatman. PicoGreen is a registered trademark of Molecular Probes, Inc. Quant‐iT is a trademark of Molecular Probes, Inc.
Products may be covered by pending or issued patents or may have certain limitations. More information.
All prices and specifications are subject to change without prior notice.
Product claims are subject to change. Please contact Promega Technical Services or access the Promega online catalog for the most up-to-date information on Promega products.