Rapid Amplification of the European Loci in a miniSTR Format
Promega Corporation
Introduction
Ever-changing demands on forensic laboratories have driven the evolution of STR technology to the next level with the PowerPlex® ESI 17 Fast (Cat.# DC1720), PowerPlex® ESI 16 Fast (Cat.# DC1620), PowerPlex® ESX 17 Fast (Cat.# DC1710) and PowerPlex® ESX 16 Fast (Cat.# DC1610) Systems. These new kits contain the same loci as in the original PowerPlex ESI and ESX Systems and meet the European Network of Forensic Science Institutes (ENFSI) and European DNA Profiling Group (EDNAP) requirements for the extended European Standard Set (ESS). The original PowerPlex® ESI and ESX Systems require 205 minutes to complete cycling and the addition of AmpSolution™ Reagent when amplifying direct-amplification samples. These new fast kits allow amplification in 50 minutes or less of both purified DNA samples and direct amplification samples such as single-source DNA immobilized on solid supports or buccal swab extracts generated with the SwabSolution™ Reagent.
The PowerPlex® ESI Fast and ESX Fast Systems were designed with the same locus configurations as the PowerPlex® ESI and PowerPlex® ESX Systems to provide the same extended ESS loci in complementary formats. The PowerPlex® ESX 16 Fast System contains the five new loci recommended by ENFSI and EDNAP (D2S441, D10S1248, D22S1045, D1S1656 and D12S391) as small amplicons and the original ESS loci (D3S1358, D18S51, TH01, vWA, D8S1179, D21S11 and FGA) as well as D2S1338, D16S539 and D19S539 as larger amplicons. This configuration is reversed in the PowerPlex® ESI 16 Fast format, with six of the seven original ESS loci as small amplicons (D3S1358, D18S51, TH01, vWA, D8S1179 and the more common FGA alleles) along with D16S539 and D19S539 as amplicons under 250bp and the five ENFSI/EDNAP loci and D2S1338 as larger amplicons (Figure 1). In addition, the PowerPlex® ESX 17 Fast and ESI 17 Fast Systems include the SE33 locus.
 Figure 1. Configurations of PowerPlex® ESX 17 Fast and ESI 17 Fast Systems.
Figure 1. Configurations of PowerPlex® ESX 17 Fast and ESI 17 Fast Systems. Panel A. The PowerPlex® ESX 17 Fast System contains the new ENFSI/EDNAP loci as miniSTRs. Panel B. The PowerPlex® ESI 17 Fast System contains the new ENFSI/EDNAP loci but focuses on miniaturization of existing ESS loci. Both configurations are available without SE33 as the PowerPlex® ESX and ESI 16 Fast Systems.
Faster Cycling with Multiple Sample Types
The PowerPlex® ESI Fast and ESX Fast Systems allow rapid amplification of both extracted DNA and direct-amplification samples with a single kit. Requiring a mere 50 minutes to amplify 500pg of extracted DNA (30 cycles) or 45 minutes for direct materials (25–27 cycles), these systems are compatible with a variety of thermal cyclers, including the GeneAmp® PCR System 9700 and Veriti® thermal cyclers (Figure 2). Concordant alleles can be generated with a variety of capillary electrophoresis instrumentation, including the ABI PRISM® 310 and Applied Biosystems® 3130 and 3500 series Genetic Analyzers (Figure 2 and data not shown). Panels and bins text files with locus-specific stutter filters are provided for data analysis with GeneMapper® ID and GeneMapper® ID-X software packages.
 Figure 2. The PowerPlex® ESX 17 Fast System is compatible with a variety of thermal cyclers.
Figure 2. The PowerPlex® ESX 17 Fast System is compatible with a variety of thermal cyclers. The thermal cycling protocol for the GeneAmp® PCR System 9700 with 30 cycles was used to amplify extracted DNA with both thermal cyclers (see the PowerPlex® ESX 17 Fast System Technical Manual # TMD040 for the detailed protocol). Amplification products were mixed with CC5 Internal Lane Standard 500 Pro (CC5 ILS Pro) and analyzed using an Applied Biosystems® 3130xl Genetic Analyzer using a 3kv, 5-second injection. Panel A. GeneAmp® PCR System 9700 results. Panel B. Veriti® thermal cycler results.
Convenience
The primer mixes contain the same autosomal STR primer sequences as the original PowerPlex® ESI and ESX Systems. The amelogenin primer pairs also are the same but have minor 5′ adjuncts to improve adenylation during fast cycling. Due to primer sequence conservation, no discordance was observed between the fast and standard cycling systems (data not shown). As with the original PowerPlex® ESI and ESX Systems, a maximum volume of 17.5µl is available for sample addition per 25µl amplification reaction. This increases the potential to recover allelic information from low-concentration DNA samples.
Robustness and Sensitivity
The PowerPlex® ESX Fast and ESI Fast Systems were compared side by side with the original standard cycling systems to gauge resistance to PCR inhibition and sensitivity. The fast systems demonstrated greater allele recovery than the standard cycling systems at high concentrations of hematin or tannic acid and comparable allele recovery at increasing concentrations of humic acid (Figure 3). Comparable recovery of alleles also was observed with the fast and standard cycling systems as the mass of DNA template decreased; there were minimal difference in the number of allele calls obtained at 30pg DNA (Figure 4).
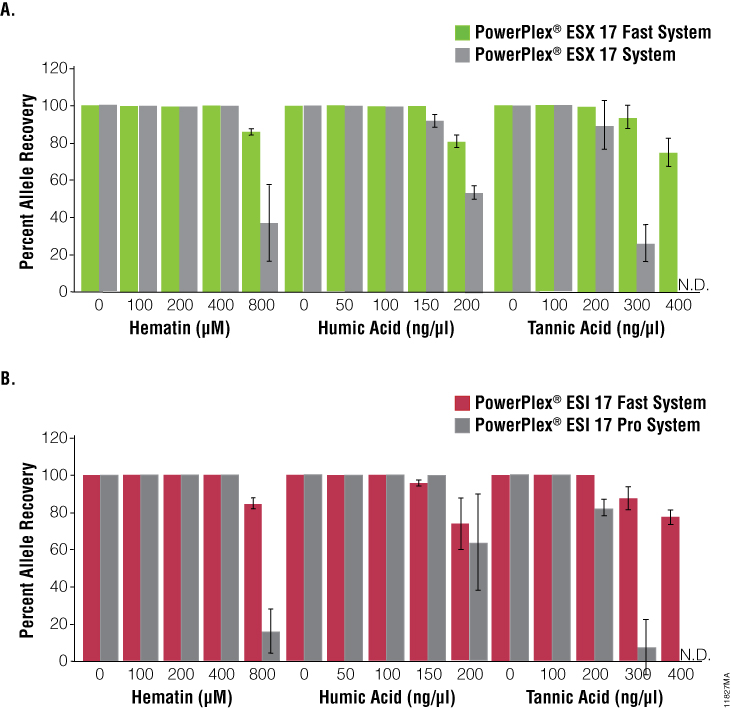 Figure 3. Performance of PowerPlex® ESX Fast and ESI Fast Systems in the presence of PCR inhibitors.
Figure 3. Performance of PowerPlex® ESX Fast and ESI Fast Systems in the presence of PCR inhibitors. Various concentrations of hematin, humic acid or tannic acid were added to amplifications with 500pg of template DNA. Panel A. Allele recovery following analysis using the PowerPlex® ESX 17 Fast and PowerPlex® ESX 17 Systems. Panel B. Allele recovery following analysis using the PowerPlex® ESI 17 Fast and PowerPlex® ESI 17 Pro Systems. N.D. = Not Detected.
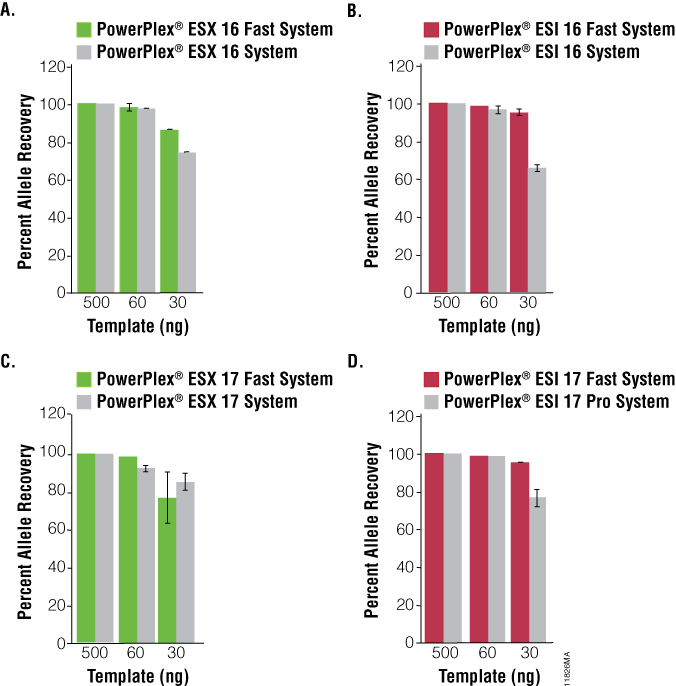 Figure 4. Sensitivity of the PowerPlex® ESX Fast and ESI Fast Systems.
Figure 4. Sensitivity of the PowerPlex® ESX Fast and ESI Fast Systems. Percent allele recovery is shown for the amplification of 500pg, 60pg or 30pg of template DNA. Panel A. PowerPlex® ESX 16 Fast and PowerPlex® ESX 16 System results. Panel B. PowerPlex® ESI 16 Fast and PowerPlex® ESI 16 System results. Panel C. PowerPlex® ESX 17 Fast and PowerPlex® ESX 17 System results. Panel D. PowerPlex® ESI 17 Fast and PowerPlex® ESI 17 Pro System results.
Direct Amplification
Direct amplification is accomplished by adding 5X AmpSolution™ Reagent (Cat.# DM1231) to the reaction at a final 1X concentration. FTA® punches may be added directly to the well, but pretreatment using the PunchSolution™ Kit (Cat.# DC9271) is required for nonFTA punches. Buccal swabs also may be used for direct amplification when SwabSolution™ Reagent is used to prepare an extract, which may be added directly to the amplification reaction. Representative results are shown in Figure 5.
 Figure 5. Direct amplification with the PowerPlex® ESI Fast and ESX Fast Systems.
Figure 5. Direct amplification with the PowerPlex® ESI Fast and ESX Fast Systems. Amplification reactions were performed in 25µl reactions containing 1X AmpSolution™ Reagent for 26 cycles. Panel A. Amplification of blood from a single 1.2mm punch from an FTA® card with the PowerPlex® ESX 17 Fast System. Panel B. Amplification of a buccal sample on an FTA® card from a single 1.2mm punch with the PowerPlex® ESI 17 Fast System. Panel C. PowerPlex® ESX 17 Fast amplification of buccal cells on an OmniSwab™ swab pretreated with SwabSolution™ Reagent. Panel D. PowerPlex® ESI 17 Fast amplification of PunchSolution™ Reagent-pretreated buccal cells collected with a Bode Buccal DNA Collector™ device. A single 1.2mm punch was added to the reaction.
Conclusions
The PowerPlex® ESI Fast and ESX Fast Systems bring all STR loci of the PowerPlex® ESI and ESX Systems to the next level with cycle times of 50 minutes or less, compatibility with purified DNA as well as direct-amplification sample types, robust performance in the presence of PCR inhibitors and comparable sensitivity with low levels of DNA template. In addition, preservation of autosomal STR primer sequences results in no discordance issues compared with the original standard cycling systems. These systems are compatible with a variety of standard thermal cyclers, with detection of amplified products on a variety of capillary electrophoresis platforms. Data analysis is supported on both GeneMapper® ID and GeneMapper® ID-X software.
Related Protocols
- PowerPlex® ESX 17 Fast System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- PowerPlex® ESI 17 Fast System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- PowerPlex® ESX 16 Fast System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- PowerPlex® ESI 16 Fast System for Use on the Applied Biosystems® Genetic Analyzers Technical Manual
- SwabSolution™ Kit Technical Manual
- PunchSolution™ Kit Technical Manual
Related Resources
How to Cite This Article
Scientific Style and Format, 7th edition, 2006
Bourdeau, J.M. et al. Rapid Amplification of the European Loci in a miniSTR Format. [Internet] . [cited: year, month, date]. Available from: https://www.promega.com/resources/profiles-in-dna/2013/rapid-amplification-of-the-european-loci-in-a-ministr-format/
American Medical Association, Manual of Style, 10th edition, 2007
Bourdeau, J.M. et al. Rapid Amplification of the European Loci in a miniSTR Format. Promega Corporation Web site. https://www.promega.com/resources/profiles-in-dna/2013/rapid-amplification-of-the-european-loci-in-a-ministr-format/ Accessed Month Day, Year.
PowerPlex is a registered trademark of Promega Corporation. AmpSolution, PunchSolution and SwabSolution are trademarks of Promega Corporation.
ABI PRISM and Veriti are registered trademarks of Applera Corporation. Applied Biosystems and GeneMapper are registered trademarks of Applied Biosystems. Bode Buccal DNA Collector is a trademark of the Bode Technology Group, Inc. FTA is a registered trademark of Flinders Technologies, Pty, Ltd., and is licensed to Whatman. GeneAmp is a registered trademark of Roche Molecular Systems, Inc. OmniSwab is a trademark of Whatman.