High-Capacity and High-Purity Antibody Purification Using Magnetic Beads
Becky Godat and Nidhi Nath
Promega Corporation
Publication Date: November 2012
Abstract
We have developed high-capacity magnetic Protein G beads (Magne™ Protein G Beads) by combining two novel technologies: magnetic beads based on macroporous cellulose and second the oriented immobilization of Protein G using HaloTag® Technology. Magne™ Protein G Beads offer an easy-to-use and rapid method for purification of polyclonal and monoclonal antibodies from variety of biological samples serum, ascites fluid and cell media. Samples from small to medium volumes can be used, and multiple samples can be purified in parallel.
Introduction
There is an increasing demand for large numbers of highly specific and sensitive antibodies for research, clinical and therapeutic use. Meeting this demand requires new tools for purifying and screening of large numbers of antibodies. Although several methods for screening large numbers of antibodies exist, including ELISAs and multichannel biosensor platforms (1) (2) (3) , high-throughput antibody purification methods have seen few developments over the years.
Antibodies are commonly purified from serum, ascites fluid and cell culture medium using Protein G‐ or Protein A‐based beads in a column chromatography format. Although well established, column chromatography has several limitations, including a need for column packing and expensive instrumentation, the inability to handle multiple samples in parallel, the inability to handle small sample volumes and the elution of a relatively dilute antibody. Magnetic bead‐based affinity purification eliminates or reduces many of these challenges and offers several advantages over conventional column‐based purification methods. Magnetic beads are easy-to-use, can process biological samples containing particulate material and do not require expensive chromatography equipment. In addition, large numbers of samples can be handled in parallel, and the purification process can be automated. Finally, it is easy to handle small amounts of beads, making it straight forward to process small sample volumes and concentrate purified antibody.
To incorporate the benefits of magnetic platforms we developed high-capacity magnetic Protein G beads, the Magne™ Protein G Beads. Here we show that these beads have several attractive features including recovery of high purity antibodies from different biological samples, concentrating antibodies and processing multiple samples in parallel.
Characteristics of Magne™ Protein G Beads
Magne™ Protein G Beads use 30 to 80 micron sized magnetic beads in which magnetic iron particles are completely covered by macroporous cellulose. These beads have very low nonspecific binding and have excellent magnetic properties making them easy to handle either manually or on a robotic platform.
A novel attachment chemistry based on HaloTag® technology is used to immobilize Protein G on the magnetic beads (4). This attachment is covalent, and the Protein G is oriented for maximum functionality. Oriented immobilization of Protein G has been shown to improve the antibody-binding capacity (5) (6) .
Magnetic Particle‐Handling Systems
To take advantage of the full potential of Magne™ Protein G Beads a variety of magnetic separation stands are available (Figure 1). Depending on which magnetic separator is used, it is possible to handle 1 to 96 samples with volumes ranging from 20µl to 50ml.

Figure 1. Magnetic separation stands that are compatible with the Magne™ Protein G Beads. Panel A. The PolyATtract® System 1000 Magnetic Separation Stand can be used with 15ml or 50ml polypropylene tubes. Panels B and C. The MagneSphere® Technology Magnetic Separation Stands are suitable for handling 1 to 12 samples in 0.5ml or 1.5ml microcentrifuge tubes. Panel D. The MagnaBot® 96 Magnetic Separation Device is for high-throughput purification using standard 96-well plates (up to 200µl sample/well) or 96-well deep-bottom plates (up to 1.0ml samples/well). Using these stands, the Magne™ Protein G Beads can be processed manually or on a robotic platform such as the Tecan for automated antibody purification.
Antibody Purification using Magne™ Protein G Beads
A typical protocol for purifying antibodies using the Magne™ Protein G Beads is shown in Figure 2. Antibodies from biological samples are captured on the beads using the strong interaction between Protein G and the Fc section of an antibody. Binding is rapid and specific and can occur in a variety of buffers with a pH range of 6.5–7.5. Bound antibody is eluted using low-pH glycine-HCl buffer (0.1M, pH 2.7) and neutralized immediately to prevent antibody inactivation.
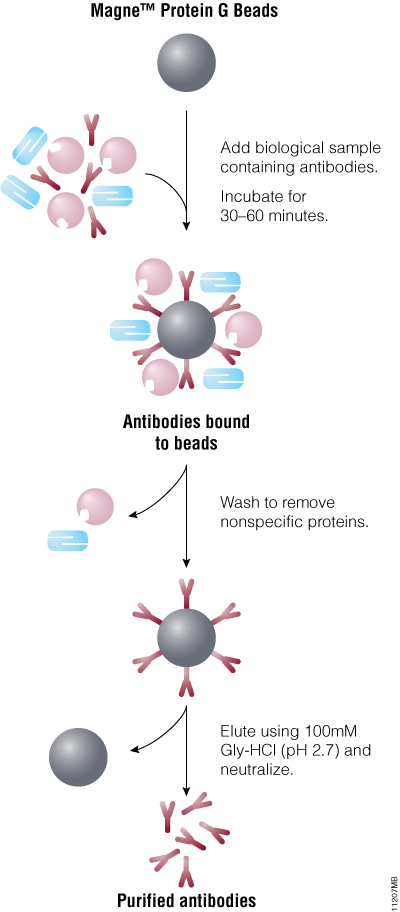
Figure 2. Schematic of antibody purification using the Magne™ Protein G Beads.
The stated binding capacity of the Magne™ Protein G Beads is greater than 18mg of human IgG per milliliter of settled beads. However, the amount of antibody purified is dependent on several factors including the biological sample, antibody isotype and antibody concentration, resulting in a measured capacity in excess of 25mg of antibody per milliliter of settled beads. Table 1 shows the amount of antibody purified from a large variety of samples containing both polyclonal antibodies and several different isotypes of monoclonal antibodies. The result highlights the sample dependent antibody purification. SDS gel analysis of purified antibodies is shown in Figure 3.
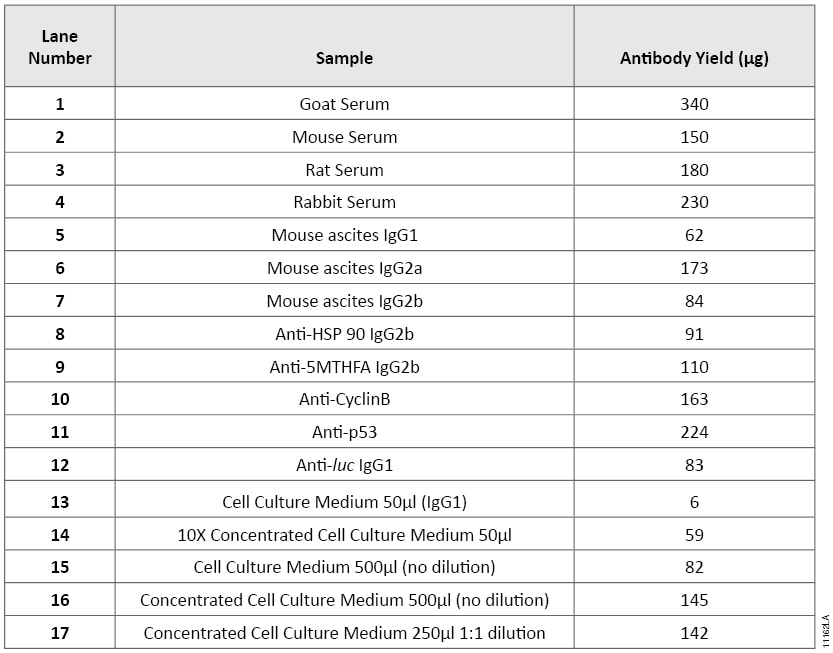
Table 1. The Amount of Antibody Purified from Different Biological Samples Using Magne™ Protein G Beads. Antibodies were purified from 50µl of each biological sample using 50µl of Magne™ Protein G Beads as described in the Magne™ Protein A Beads and Magne™ Protein G Beads Technical Manual #TM371. Sample 8, 9, 11 and12 are purified monoclonal antibodies purchased commercially. Sample 10 is an IgG polyclonal antibody. Samples 13–17 are cell culture medium expressing mouse IgG1. Medium was concentrated tenfold (samples 14, 16 and 17) using a filter with a molecular weight cutoff of 3,000 Daltons.
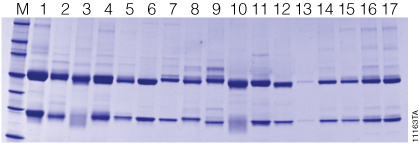
Figure 3. SDS gel image of 17 antibody samples purified using Magne™ Protein G Beads. Lane M. Broad Range Protein Molecular Weight Markers (Cat.# V8491). Sample details are given by lane number in Table 1. Each lane contains 2.5µl of sample in SDS loading buffer. The samples were heat denatured at 95°C for 5 minutes and then loaded on 4–20% tris-glycine gel. SimplyBlue™ safe stain was used to visualize the gel.
Optimization of Antibody Purification
Depending on the intended downstream application, several different purification parameters can be optimized including the elution conditions and the amount of Magne™ Protein G Beads. For example, Figure 4 shows the result of a study where Human IgG bound to the Magne™ Protein G Beads is eluted in two fractions and the antibody recovery is calculated for each fraction. The results show that the majority of antibody is recovered in the first elution fraction, and this antibody is at a high concentration. Additional antibody is recovered in the second fraction at a lower concentration. In addition, varying the elution times shows that reducing the elution time from the recommended 5 minutes only results in a small decrease in antibody recovery (Figure 4). The ability to reduce elution times can be important for antibodies that may not be stable during prolonged exposure to low pH conditions.
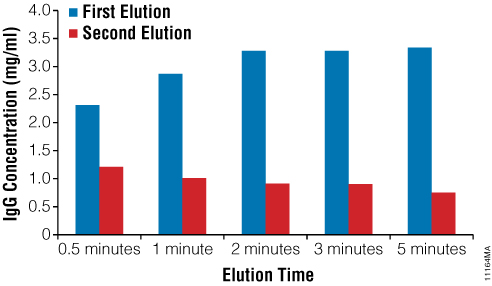
Figure 4. Antibody elution profile as a function of time and number of elutions. Human IgG (500µg) was purified using 50μl of Magne™ Protein G Beads as described in the Magne™ Protein A Beads and Magne™ Protein G Beads for Antibody Purification Technical Manual #TM371. Bound antibody was eluted in two fractions at the indicated elution times using 50μl of 100mM Glycine‐HCl buffer (pH 2.7) and immediately neutralized with 10μl of 2M Tris‐HCl (pH 7.5). Antibody concentration in each fraction was determined using absorbance at 280nm.
The amount of Magne™ Protein G Beads can be optimized if high antibody recovery is important. Figure 5 shows that increasing the amount of Magne™ Protein G Beads from 25–200µl of bead slurry increases the antibody recovery from 30% to almost 80% of the total input amount.
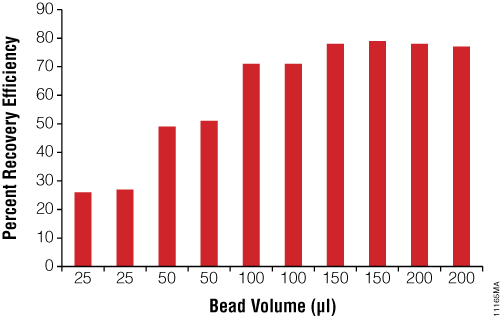
Figure 5. Antibody recovery as a function of Magne™ Protein G Bead slurry. Samples containing 500µg of Human IgG was purified using increasing amount of Magne™ Protein G Beads following the Magne™ Protein A Beads and Magne™ Protein G Beads for Antibody Purification Technical Manual #TM371.
Antibody purification can be optimized as a function of antibody concentration, antibody isotype and amount of Magne™ Protein G Beads (Figure 6). The results show that a sample with antibody concentration as low as 2.0µg/ml can be purified using Magne™ Protein G Beads. Although the amount purified is dependent on the antibody isotype. As shown before, most of the antibody is eluted in the first fraction with an additional amount eluting in the second fraction at lower concentration. No significant improvement is seen when the amount of Magne™ Protein G Beads is increased from 2µl to 50µl. Similar optimization would be needed for other sample types. However, because the Magne™ G Beads are easy to handle, several variables can be tested in parallel using small sample volumes (e.g., in a 96-well format).
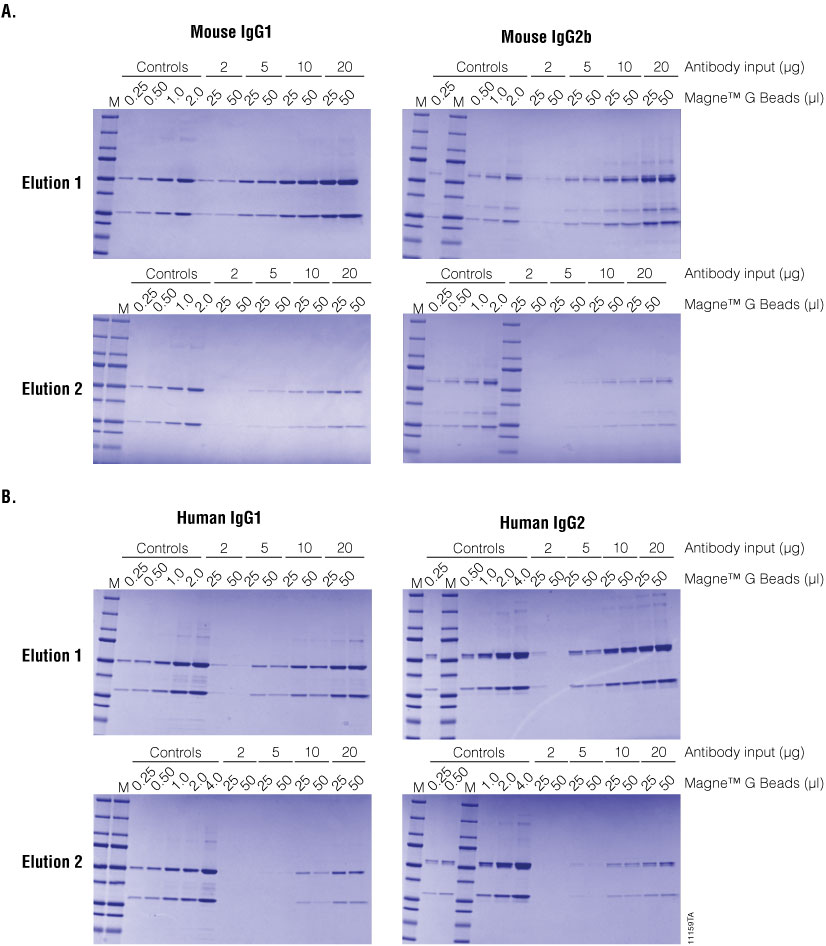
Figure 6. Antibody recovery as a function of Magne™ Protein G Bead volume. Antibody purification from PBS buffer spiked with 2, 5, 10 or 20µg/ml of human IgG1, human IgG2, mouse IgG1 or mouse IgG2. Panel A. Mouse IgG1 and mouse IgG2b. Panel B. Human IgG1 and human IgG2. For all samples, 1ml was processed using 25µl or 50µl of Magne™ Protein G Bead slurry as described in the Magne™ Protein A Beads and Magne™ Protein G Beads for Antibody Purification Technical Manual #TM371.
Manual Purification of Antibody from Multiple Samples Using Magne™ Protein G Beads
A key advantage of Magne™ Protein G Beads over traditional column‐based antibody purification is the ability to perform small‐scale antibody purifications using 96‐well plates. The magnetic properties of the Magne™ Protein G Beads allow them to be manually manipulated using a magnetic separation device such as the Deep Well MagnaBot® 96 Magnetic Separation Device (Cat.# V3031). Table 2 shows the results of using the Deep Well MagnaBot® 96 Magnetic Separation Device to purify antibody from 50μl of goat serum using 50μl of Magne™ Protein G Beads. All samples yielded excellent antibody recovery with minimal well‐to‐well variation.
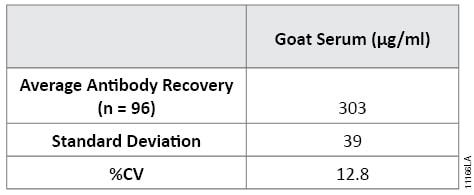
Table 2. Manual High-Throughput Antibody Purification using Magne™ Protein G Beads with the Deep Well MagnaBot® 96 Magnetic Separation Device.
Summary
parallel. The amount of antibody purified is dependent on antibody isotope, antibody concentration, sample volume and amount of Magne™ Protein G Beads. Because of the excellent handling properties of the Magne™ Protein G Beads optimization experiments can be multiplexed at a small scale for rapid results. Once optimized, the purification protocol can be used to process large numbers of samples in parallel or scale up using tested protocols for samples up to 50ml. Access to purified antibodies allows researchers to perform critical antibody screening and characterization early in the clone selection process and improves the chances of picking up the right antibodies.
The Magne™ Protein G Beads contain high-capacity antibody purification magnetic beads that offers a convenient method to purify antibodies from a variety of biological samples. Unlike column chromatography, the Magne™ Protein G Beads, in conjunction with a wide variety of magnetic particle handling systems, can be used to handle larger numbers of samples in
References
- Säfsten, P. et al. (2006) Screening antibody-antigen interactions in parallel using Biacore A100. Anal. Biochem. 353, 181–90.
- de Wildt, R.M. et al. (2000) Antibody arrays for high-throughput screening of antibody-antigen interactions. Nat. Biotechnol. 18, 989–94.
- Ayriss, J. et al. (2007) High-throughput screening of single-chain antibodies using multiplexed flow cytometry. J. Proteome Res. 6, 1072–82.
- Los, G.V. et al. (2005) HaloTag™ Interchangeable Labeling Technology for cell imaging, protein capture and immobilization. Promega Notes 89, 2–6.
- Jung, Y. et al. (2007) Self-directed and self-oriented immobilization of antibody by protein G-DNA conjugate. Anal. Chem. 79, 6534–41.
- Lee, J.M. et al. (2007) Direct immobilization of protein g variants with various numbers of cysteine residues on a gold surface. Anal. Chem. 79, 2680–7.